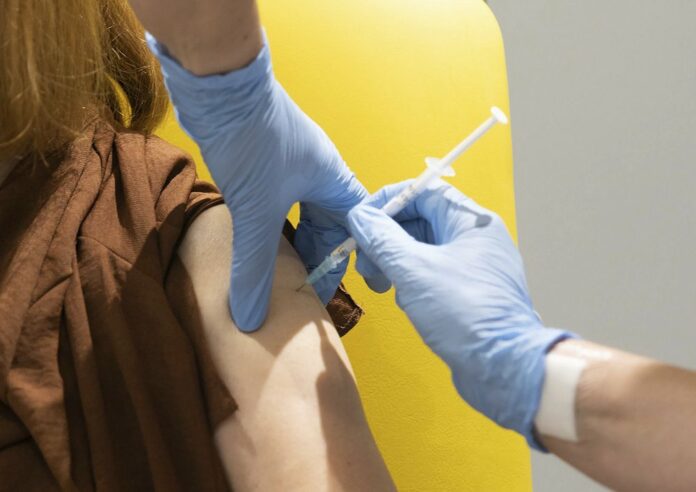
BANGKOK — British pharmaceutical AstraZeneca became the first company to receive approval from the Food and Drug Administration to sell and distribute coronavirus vaccine for domestic use in Thailand.
In an announcement made on Thursday evening, FDA sec-gen Paisarn Dunkum said the approval was granted to AstraZeneca on Wednesday after deliberating on more than 10,000 pages of documents filed by the British company.
The decision means that Thai companies and hospitals can now import vaccines from AstraZeneca for their own uses, though Paisarn said officials from the Department of Medical Sciences will conduct a random test on the doses once they arrive in Thailand before they can be handed out to the public.
Paisarn added that the first shipment of vaccine from AstraZeneca is due to arrive “in February,” consisting of 50,000 doses. More shipments, containing up to 150,000 doses in total, will follow in March and April, he said.
Millions of doses will also be manufactured locally by Siam Bioscience, a company wholly owned by the palace, per an agreement with AstraZeneca, according to the government.
Paisarn estimated that the first batch of coronavirus vaccine made by Siam Bioscience will be available by May.
The FDA has yet to grant an approval for the vaccine developed by Chinese pharmaceutical Sinovac. Officials have previously said 2 million doses from Sinovac will be distributed to frontline health workers and vulnerable populations in the five provinces most affected by the new wave of the coronavirus outbreak.
Related stories:
Thais Most Willing to Take Vaccine, Out of 24 Surveyed Countries