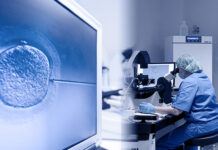
Top: A researcher on July 4, 2019, inspects a marijuana plant cultivated by a state agency for research purposes. Image: Government Pharmaceutical Organization.
Reopening its cannabis regulatory platform after months of disruption caused by the coronavirus, the Food and Drug Administration has prepared a draft bill to abolish a 5-year ban on the private sector making cannabis medicine.
Two draft ministerial regulations, one on marijuana and the other on hemp, under scrutiny since the end of last year and January respectively by the Council of State—the government’s legal arm—are also inching ahead following the easing of the lockdown.

Under these three pieces of legislation and regulations, foreigners will be allowed to get involved as long as they hold shares not exceeding one-third in a company incorporated under local law.
International travelers are the most relaxed category and are always eligible to apply for import and export licenses to bring in and take out cannabis medicine to treat their illnesses.
The Ban
The present cannabis law sets out qualifications of an applicant for a production, import or export license to be limited only to state agencies or state-agency backed farmer groups and private universities.
Non-state entities are not eligible to apply.
The law also forbids companies from applying for a license to grow cannabis plants or manufacture cannabis medicine from the date the law came into effect on February 19, 2019 until five years later, ending on February 19, 2024.

As a result, there has been an acute shortage of supply of medicine containing CBD—the cannabidiol chemical compound extracted from the flower of marijuana and hemp.
Patients that would like to be treated by CBD oil have no access to the medicine and hundreds of cannabis sale licenses granted to doctors, dentists and pharmacists have been rendered useless as there are no drugs to prescribe.
A Partial Repeal
One disappointing aspect of the proposed legislation is that it lifts the production ban only in relation to Thai traditional medicine and herbal cannabis.
Modern CBD pharmaceuticals continue to be banned.
What this persistent ban entails is that those companies that plan to produce modern medicinal CBD oil are not qualified as an applicant themselves to apply for a production license, either to grow or to manufacture cannabis, and have to pair up with a state entity to seek a license on their behalf.
This way, the potential of companies being able to make modern cannabis medicine remains in an indirect path around cooperation with a state agency.
The proposed new law would enable companies to directly apply for a license to agriculturally grow marijuana and hemp plants and industrially manufacture CBD oil for medicinal purposes as long as the products are categorized as Thai traditional medicine or herbal goods.
Another requirement is for the applicant to court cooperation and supervision from an existing licensee of modern medicine, Thai traditional medicine or herbal cures under the separate pharmaceutical law.
Foreign Patients Exempted
To protect the Thai economy, understandably, imports of cannabis remain restricted.
The 5-year ban will continue to be upheld for imports of cannabis and hemp products, with the exception of importing seeds from overseas to be used for growing marijuana and hemp plants in agriculture.
But foreign patients who need medical cannabis are not affected. Under both the current and new laws, foreign individuals who need cannabis medicine to treat their ailments are well protected.

International travelers will be free to carry with them or otherwise import into Thailand their own CBD oil after applying for and receiving an import license.
To the jubilation of the masses and foreigners residing in the kingdom, those who have been certified as patients by professional medical practitioners would be permitted to grow cannabis for their own treatment.
This is the government’s effort to universally widen access to affordable cannabis health care.
Push for Cannabis Exports
In a show of the country’s ambition, long anticipated since the start of the cannabis legalization, exports of cannabis and hemp as plants, parts of plants and as finished products, modern and traditional or herbal, are totally liberated by the proposed law from the 5-year ban.
Once an export license has been obtained, a licensed cannabis producer will be able to ship their cannabis- and hemp-based modern CBD medicine and herbal products to the world.
So will a non-producer export license holder.

International travelers, too, will be able to apply for an export license to take their CBD treatments out of the country.
The draft legislation is taking a long journey from the FDA to the health ministry and to the Cabinet, then to the Council of State and back to the Cabinet before being debated and enacted in Parliament.
Realistically, with the pandemic still pretty much a worry, the proposed bill is unlikely to become law until sometime in 2021.
Hemp Goes Beyond Medicine
Hemp regulation, once a promising move, has also been victimized by the coronavirus outbreak.
This is unfortunate because hemp (Cannabis sativa L. subsp. sativa) offers a bright hope for the government to turn it into an economic crop beyond CBD oil and medicine.
The draft ministerial regulation on hemp currently in the hands of the Council of State spans a wide range of uses from medicine to household consumption to edibles, food, drinks and cosmetics.
The draft regulation was released by the Cabinet at the beginning of this year to the government’s career legislation experts at the Council of State, and then COVID-19 struck.
There will be three readings at the organization before the regulation is sent back to the Cabinet and the health ministry to be signed into law. The first reading is underway.
This regulation will be prioritized higher than the draft regulation on cannabis (Cannabis sativa L.), which had arrived at the council earlier towards the end of last year and already passed the first reading.
With the shutdown eased, cannabis and hemp law and regulations are finally moving ahead with a vengeance.
Wirot Poonsuwan is the Senior Counsel and Head of Special Projects at Blumenthal Richter & Sumet in Bangkok and can be reached at [email protected].